Duchenne/Becker Muscular Dystrophy Clinical Trials
Now Recruiting
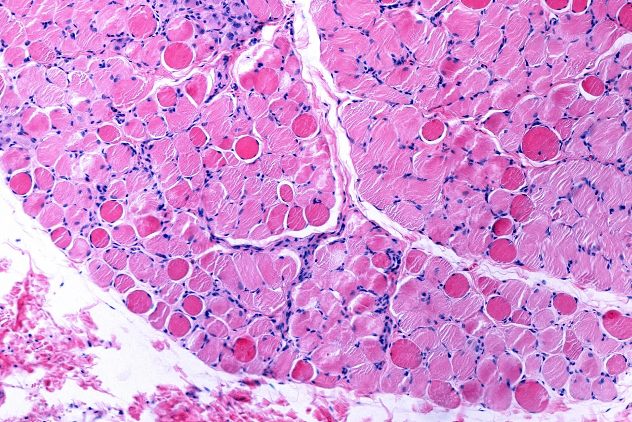
Duchenne and Becker muscular dystrophies are rare, genetically inherited neuromuscular disorders that lead to progressive muscle weakness and degeneration, profoundly affecting individuals and families across the world. At the heart of both conditions is a mutation in the DMD gene, which provides instructions for making and regulating dystrophin, a protein essential for stabilizing and protecting muscle fibers during movement and everyday activity. In Duchenne muscular dystrophy (DMD), mutations typically prevent the body from producing functional dystrophin, causing symptoms to emerge in early childhood and leading to loss of ambulation and serious complications involving respiratory and cardiac function over time. In contrast, Becker muscular dystrophy (BMD) results in the production of partially functional dystrophin, which usually leads to a later onset and slower progression of muscle weakness.
At the Neurology Rare Disease Center, we are deeply committed to advancing therapeutic options and improving lives through clinical research. Clinical trials play a vital role in understanding the natural history of these diseases, evaluating new treatments, and expanding the possibilities for disease-modifying therapies. Participation in clinical trials not only contributes to medical breakthroughs that may benefit future generations, but also gives patients and families access to cutting-edge care and expert support every step of the way.
We encourage you to connect with our experienced team to explore the clinical trial opportunities available and learn about eligibility. Together, we can strive toward better outcomes, heightened hope, and transformative advances in treatment.

